All News
All news
-
News BIOCORP SIGNS AN INDUSTRIALIZATION CONTRACT WITH VIRBAC
BIOCORP signs an industrialization contract with Virbac for the manufacturing of an innovative administration and closure system compatible with vials. -
News FDA Advisory Committee to review Avastin biosimilar candidate
The Committee will review analytical, pharmacokinetic and clinical data from studies involving ABP 215, including results from a Phase III study in patients with NSCLC. -
News Merck's SOLu-Trypsin enzyme brings stability to mass spectrometry
Using the solution stable enzyme can eliminate unnecessary waste and cost. -
News Novatek International selected as partner for automation of Boehringer Ingelheim's global environmental monitoring program
NOVA-EM is Novatek's solution dedicated to managing all aspects of an automated environmental monitoring program. -
News TraceLink unveils Automated Validation Manager
Removes burden of validating software updates for Life Sciences Cloud customers. -
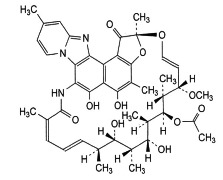
News New BMS collaboration to evaluate combination therapy in colorectal cancer
Phase I/II study to evaluate the combination of Opdivo (nivolumab) and Opdivo +Yervoy (ipilimumab) regimen with Mekinist (trametinib) in patients with colorectal cancer. -
News Pharmaceutical contract manufacturing/contract research market worth $238.3 billion by 2025
Expanding market for biosimilars and biobetters is anticipated to pronounce the demand for contract development and manufacturing support. -
News Lonza acquires cell and gene contract manufacturer PharmaCell
Lonza has successfully completed the acquisition of PharmaCell, a cell and gene contract manufacturer in Europe with employees in Maastricht and Geleen (NL). In 2016, PharmaCell had sales of EUR 11 million. -
News Acasti Pharma and CordenPharma announce large-scale production of CaPre with continuous manufacturing
Continuous manufacturing process will enable improved quality control and cGMP compliance, while reducing energy consumption, waste and raw material. -
News Colorcon expands in India
The new technical service laboratory is the company's 20th across the world. -
News Combination therapy shown to shrink tumours in many patients whose cancer has spread to the brain
Combination of nivolumab plus ipilimumab can fundamentally change survival expectations. -
News Sartorius Stedim Biotech launches ambr 15 bioreactor system with Nova BioProfile FLEX2 integration
QbD studies in upstream processing more rapidly performed. -
News Novartis announces ground-breaking collaboration with IBM Watson Health on outcomes-based care in advanced breast cancer
Collaboration will use real-world patient data and cognitive computing with aim of improving outcomes in advanced breast cancer. -
News Gen-Plus
Gen-Plus is a small medium-sized, privately owned company.
Its aim is to discover innovations for contract partners. We
design formulation and technology concepts for the
pharmaceutical industry. This includ... -
News Spinraza approved in the EU as first treatment for spinal muscular atrophy
Robust data from Phase III studies demonstrated positive impact on motor milestone achievement; increased survival in infants with SMA. -
News Extractables-free deep-well plate for uHPLC and MS applications
Porvair Glass Vial Deep-well plates are precisely manufactured to ensure complete compatibility with automated equipment. -
News NICE approves first use of Opdivo in blood cancer
NICE has recommended immunotherapy drug nivolumab for the treatment of adult patients with classical Hodgkin lymphoma. -
News Vertex reaches long-term reimbursement agreement with ROI for Orkambi, Kalydeco and future CF medicines
Agreement provides access to Orkambi for people who have two copies of the F508del mutation and expands access to Kalydeco for all eligible patients. -
News NIH researchers find potential genetic cause of Cushing syndrome
Finding may lead to therapies that prevent pituitary tumor recurrence. -
News Cardinal Health Specialty Solutions publishes research-based insights on oncology
Pharmaceutical companies should play a larger role in patient support programs. -
News Hikma signs an exclusive license and supply agreement with Octapharma for Octaplex
Deal adds the first plasma-derived treatment to Hima's product offering. -
News Sandoz proposed biosimilars adalimumab and infliximab accepted for regulatory review by the EMA
Biosimilar infliximab alone could potentially save the NHS £89 million. -
News Vetter and Microdermics to develop innovative microneedle drug delivery systems
Microdermics has successfully demonstrated the initial safety of its microneedle system, and is planning Phase I human clinical trials for vaccine and therapeutic delivery, to be initiated in 2017. -
News Synimmune initiates FiH study of Fc-optimized antibody Flysyn for the treatment of AML
Flysyn is the first antibody from the company's pipeline to be tested in humans. -
News Cambrex invests in new capacity and continuous flow technology at its Karlskoga, Sweden facility
The expansion is part of an ongoing strategic campaign to invest in small molecule API manufacturing across the company's global network of facilities. -
News Sanner acquisition strengthens company's leadership in effervescent packaging market
Acquisition of Jaco enables Sanner to operate as a system supplier. -
News Orchard Therapeutics signs manufacturing services agreement with PCT Cell Therapy Services
Under the terms of this new agreement, PCT will provide GMP-compliant manufacturing services for Orchard’s lead product, OTL-101, an autologous ex-vivo gene therapy for the treatment of ADA-SCID. -
News Hookipa Biotech announces data showing TheraT turns cold tumours hot
TheraT mediated alarmin release crucial for active immunization in cancer immunotherapy. -
News Recipharm makes strategic appointment in the UK & Ireland
Shabbir Mostafa to join the CDMO as Director, Business Management, with responsibility for driving sales of Recipharm’s manufacturing services. -
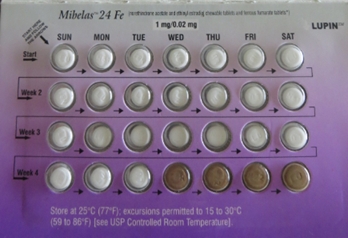
News Lupin Pharmaceuticals announces nationwide recall of Mibelas 24 Fe tablets
Recall due to out of sequence tablets and missing expiry/lot information. -
News Marken Launches Hybrid Clinical Trials Service
Marken today announced it has launched a new hybrid logistics service which leverages the UPS global transportation network, and has already received customer commitments for the new service. The hybrid service enables Marken to utilize the UPS network... -
News FDA accepts for priority review BMS’s application for Opdivo in previously treated hepatocellular carcinoma
Application is based on results from the Phase I/II CheckMate -040 trial. -
News Inovio HIV vaccine elicits nearly 100% immune response rates in a clinical study
Amongst highest levels of immune responses ever demonstrated in an HIV vaccine human study. -
News Pharmaceutical case studies illustrating data analytics strategies
New webinar hosted by Xtalks focuses on extracting the most value from data assets. -
News PDA initiative connecting drug manufacturers, glass suppliers to make manufacturing great
First initiative is to eliminate visible particles from parenteral products, -
News Innovation in packaging is here!!
Unipharma has applied state-of-the-art technology to traditional products and made the first ever Mono-Dose/Unit-Dose OTC line of products which will forever change the way people consume medications. No more confusions, no more charts, no more scie... -
News Merck strikes deal with Teijin Pharma for investigational antibody candidate targeting tau
The addition of this antibody targeting tau will complement Merck’s portfolio of candidates being investigated for the treatment of Alzheimer's disease. -
News WuXi STA, CFDA and Jinshan Government to co-host Marketing Authorization Holder regulatory summit in Shanghai
Industry and regulators combine to run afternoon congress on the implications of regulatory changes. -
News Hovione starts clinical trial of its proprietary minocycline sterile ointment to treat a subset of anterior ocular inflammation
First clinical study using a patent protected minocycline API and formulation developed by Hovione. -
News PharmaMar and Eczacıbaşı sign a licensing agreement for Aplidin in Turkey
Aplidin is PharmaMar's second most advanced anticancer drug currently under development for the treatment of multiple myeloma and angioimmunoblastic T-cell lymphoma. -
News IPF patients treated with Ofev vs placebo twice as likely to have improved or stable lung function
Separate subgroup analysis demonstrates long-term efficacy of Ofev in slowing disease progression is maintained for up to 96 weeks in IPF patients who require dose adjustments to manage adverse events. -
News Bioverativ to acquire clinical-stage rare disease biotechnology company, True North Therapeutics
Strengthens pipeline with lead candidate TNT009 in cold agglutinin disease, a rare and chronic autoimmune hemolytic anemia with no approved therapies. -
News OPKO R&D Facility
EirGen have commenced construction of a state of the art R&D facility in the Business and technology Park, Cork road, Waterford (Opposite the Whitfield clinic). The facility will be construction complete in Q2 2018 with a phased start up leading to... -
News New Rotogravure printing machine.
New rotogravure printing machine and a new cutting machine in operation since the end of 2014. -
News FDA approves Kevzara for the treatment of moderately to severely active RA in adults
Kevzara is now available to US patients. -
News New analyses reinforce the potential of Ultibro Breezhaler for COPD patients historically treated with steroids
New analyses from the FLAME study suggest dual bronchodilator Ultibro Breezhaler provides similar or better efficacy versus steroid-containing therapies, regardless of blood eosinophil counts. -
News Spiriva Respimat inhalation spray improves breathing for people with asthma regardless of BMI or allergic status
In all analyses, people experiencing uncontrolled asthma symptoms saw improvements in their breathing by adding Spiriva Respimat to an ICS or combination ICS/LABA. -
News FDA approves Genentech’s Actemra for giant cell arteritis
Sixth FDA approval for Actemra since its US launch in 2010, -
News Thermo Fisher Scientific to acquire Patheon
Patheon provides entry into the attractive, high-growth CDMO market. -
News Boehringer Ingelheim inaugurates biopharmaceutical manufacturing facility in China
It is the first and only biopharmaceutical manufacturing site established by a multinational active pharmaceutical company in China. -
News Hikma launches generic version on Edecrin in the US market
Product adds to the company's non-injectables portfolio in the US. -
News EMA validates application for BMS’s Sprycel in children with chronic myelogenous leukemia
Proposal extends application to the treatment of children and adolescents with chronic phase Philadelphia-chromosome positive chronic myelogenous leukemia and to the powder for oral suspension. -
News Biogen acquires Remedy Pharmaceuticals’ Cirara for large hemispheric stroke
Phase III-ready program complements Biogen’s ongoing development efforts in stroke. -
News Irvine Scientific launches new xeno-free medium for hematopoietic progenitor cell culture
New medium designed for hematopoietic cell culture for cell-based therapies. -
News Merck's CHOZN expression system selected for bi-specific antibody development by SystImmune
Accelerates development timelines with faster, easier selection and scale up of clones. -
News CPHI Worldwide to launch global pharma market ranking index
Research used to produce a global ranking of countries’ pharma reputation, market growth potential, innovation and competitiveness. -
News SGS expands stability capacity at Mississauga, Canada laboratory
Expansion part of global investment in testing capabilities. -
News Sartorius Stedim Biotech partners with Nova Biomedical
Unique tool will allow massive quantities of cell culture data to be collected during upstream processing QbD studies. -
News Saneca Pharma receives confirmation of multi-dosage cGMP approval for Russia
Certification covers the manufacture and packaging of hard and soft gel capsules, liquids for external use, semi-solids such as ointments and film coated tablets. -
News Friulchem newsletter Apri 2018
Exclusive worldwide license for FC-Cubes -
News Friulchem S.p.A. Issued Patent for Rifaximin
Friulchem S.p.A., a chemical-pharmaceutical company active in human and veterinary fields, has received an US Patent (US Patent 14/11856) covering own solid form called “pseudo-crystalline” solid form, derived from Rifamycin O.
-
News Gamlen Tableting changes its name to Gamlen Instruments
New company name reflects diverse market opportunities. -
News Aptar Pharma’s electronic Lockout device approved by EMA
The e-Lockout device being the first and only fully integrated electronic nasal drug delivery device to be approved by a US or European regulatory authority. -
News Cambrex invests to increase pilot-scale API capacity at its High Point, NC facility
Increases site’s reactor capacity by 30%. -
News SGS Expands Stability Capacity at Mississauga Canada Laboratory
SGS has expanded storage capacity at its laboratory in Mississauga, Canada, for the effective stability testing of new drug substances and products for full regulatory compliance. -
News Catalent and Therachon to advance clinical development of innovative protein therapy for achondroplasia
Catalent Biologics will use its proprietary GPEx technology to produce different protein variants for Therachon, -
News Merck launches industry's first off-the-shelf cell culture media for perfusion processes
Allows customers to achieve a more optimal output than they would using conventional batch or fed-batch processes. -
News Research reveals manufacturers and contract packagers may miss serialization deadlines
Many companies currently unprepared because they lack the internal resources to devote to serialization. -
News Genentech to present new data on personalized medicines and cancer immunotherapies
New pivotal results for Perjeta and Alecensa demonstrating improvement over standards of care. -
News Avastin as effective as Eylea for treating central retinal vein occlusion
NIH-funded clinical trial shows use of either drug improved vision and had few side effects. -
News Hikma receives CRL regarding its ANDA for generic Advair Diskus
Company believes low likelihood of approval this year. -
News Merck's Keytruda scores another FDA approval
FDA approves Keytruda as first-line combination therapy with pemetrexed and carboplatin for patients with metastatic nonsquamous non-small cell lung cancer (NSCLC) irrespective of PD-L1 expression. -
News Takeda terminates plans for Vaxem Hib launch in Japan
Discontinuation due to the vaccine supplier’s (GSK) decision to discontinue global production. -
News New licensing program facilitates adoption of Tandem Mass Tag technology
Thermo Fisher Scientific helps research service providers and their customers optimize protein identification and quantitation. -
News Croda celebrates EXCiPACT Certification across all leading excipient sites
Status marks the company as the first multi-site excipient supplier to achieve global EXCiPACT accreditation across its production facilities. -
News Gyros Protein Technologies introduces next generation Gyrolab Protein A Kit for biotherapeutics
Kit increases efficiency of residual protein A ligand detection. -
News Roche's Tecentriq flops during Phase III trial
IMvigor211 failed to meet its primary endpoint of overall survival (OS) compared with chemotherapy. -
News CPHI Worldwide Advisory Board returns with five new global experts
Board helps to steer the overall development of CPHI events and content. -
News Optibrium and Goldfinch Bio sign agreement for StarDrop license
Goldfinch Bio’s researchers select StarDrop to guide successful drug discovery. -
News Catalent provides commercial manufacturing of Lexicon Pharmaceuticals' orphan drug Xermelot following FDA approval
Xermelo is a first-in-class orally administered therapy for the treatment of carcinoid syndrome diarrhea. -
News Bosch Packaging Technology presents Xelum platform for continuous manufacturing
Highly precise dosing of smallest amounts of API. -
News New buprenorphine wafers could help thousands battling opioid addiction
When compared with current hard sublingual tablets, the novel formulation dissolved quickly with no increased safety concerns and no demonstrable difference in efficacy. -
News Cross-industry partnership reduces the cost of popular diabetes, asthma and other brand name drugs
Inside Rx, a new initiative from Express Scripts, partners with GoodRx, drug manufacturers and drug stores to save Americans an average of 34% at 40,000 pharmacies -
News First approval for Keytruda in a hematologic malignancy in the EU
European Commission approves Keytruda for patients with relapsed or refractory classical Hodgkin lymphoma who failed ASCT and BV or who are transplant-ineligible and have failed BV. -
News Cherwell Laboratories to highlight cleanroom microbiology solutions at The Clinical Pharmacy Congress
A key focus on Cherwell’s Stand A49 will be the microbiology requirements for pharmacy aseptic manufacturing, preparation and dispensing. -
News ProSolus doubles transdermal drug delivery manufacturing capacity
New ProSolus commercial team will provide potential partners throughout the pharmaceutical industry direct access to the state-of-the-art research, development, and manufacturing capabilities. -
News SGS Clinical Research announces results of FiH studies using novel virus as experimental challenge agent
Strain represents one of the common current circulating strains of influenza of a pandemic origin. -
News Arch Biopartners starts GMP manufacturing for AB569 at Dalton Pharma Services
Company will be responsible for the GMP preparation and filling of AB569 into glass vials. -
News Sartorius Stedim Biotech launches chemistry testing services
This chemistry testing offer complements the company’s existing range of off-the-shelf biological assays for biosimilar products, providing a complete solution from one contract partner. -
News SGS Announces Results of First-In-Human Studies Using Novel Virus as Experimental Challenge Agent
SGS announced the successful completion of a Phase I clinical trial of a new strain of influenza virus in healthy volunteers. -
News Aptar Pharma partners with Kali Care to develop real-time medication management technology
Combined expertise is likely to have a significant impact in reducing the costs and complexity of ophthalmic clinical trials. -
News NIH discovery in mice could lead to new class of medications to fight mid-life obesity
With lower rates of obesity, the researchers say rates of heart disease, diabetes, and other diseases that tend to increase with age, including cancer and Alzheimer’s disease, could fall as well. -
News Isogenica introduces next-generation, fully synthetic human Fab antibody fragment library
Library claimed to offer "the most efficient route to therapeutic antibody lead identification". -
News Native Antigen Company expands into new development and production facilities
Expansion doubles manufacturing capacity and includes the introduction of new facilities. -
News AstraZeneca’s Imfinzi receives FDA accelerated approval for previously treated patients with advanced bladder cancer
Imfinzi is the cornerstone in an extensive immuno-oncology program across multiple cancer types and stages of disease. -
News Concert Pharmaceuticals initiates CTP-543 Phase II trial in alopecia areata
Trial designed to evaluate the safety and efficacy of CTP-543 after 12 months of dosing with the primary efficacy analysis at week 24. -
News GSK invests $139 million to expand production capacity for Benlysta in Rockville
Site is also expected to house production of a new subcutaneous form of belimumab, which is currently under review with the FDA. -
News Shire and Parion Sciences sign collaborative license agreement to advance P-321 for ophthalmic indications
P-321 is a Phase II investigational topical treatment for dry eye disease. -
News Impax announces FDA approval and launch of a generic version of Vytorin
One of the first companies to offer a generic version of Vytorin. -
News Takeda announces FDA Accelerated Approval of Alunbrig
Alunbrig approved for ALK+ metastatic non-small Cell lung cancer patients who have progressed on or are intolerant to crizotinib. -
News China FDA approves country’s first all-oral regimen for chronic hepatitis C, Daklinza in combination with Sunvepra
Daklinza and Sunvepra combination approved for genotype 1b, the most common chronic hepatitis C (HCV) genotype in China; combination has a 91-99% cure rate. -
News Sartorius introduces new BIOSTAT STR bioreactor range and new Flexsafe STR single-use bags
New generation of bioreactors combined with new Flexsafe STR bags offer a fully scalable, single-use system. -
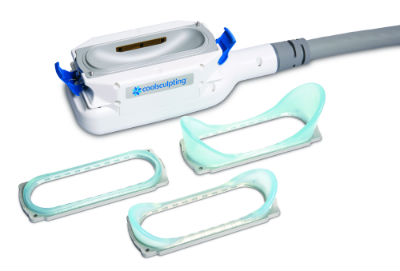
News Allergan successfully completes ZELTIQ Aesthetics acquisition
Acquisition adds best-in-class body contouring business to Allergan's facial aesthetics, plastic surgery and regenerative medicine businesses. -
News Record growth for life science tech firm as MES adoption increases globally
Zenith Technologies surpasses the €20 million milestone for sales of its manufacturing execution system (MES) services. -
News Learn from change-making pharmaceutical leaders: CPHI North America keynote speakers announced
CPHI North America will feature the first-ever Connect Conference. -
News Antidepressant may enhance drug delivery to the brain
NIH rat study suggests amitriptyline temporarily inhibits the blood-brain barrier, allowing drugs to enter the brain. -
News I Holland offers free trial of its innovative tool management system
System allows tablet manufacturers to keep a record of tablet quantities by number of tablets, work order or batch information to ensure production is run efficiently. -
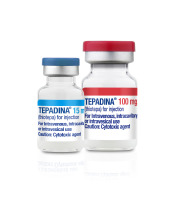
News First FDA-Approved 100 mg Thiotepa Now Available through Amneal Biosciences as TEPADINA® (thiotepa) for Injection
Thiotepa is now available through Amneal Biosciences in both 100 mg/vial and 15 mg/vial as branded TEPADINA® (thiotepa) for injection. The product is supplied as a powder for solution in single-dose vials for intravenous, intracavitary, or intravesical... -
News Pharmatek adds further cGMP spray drying capacity to meet demand for improved pharmaceutical solubility
Company has also installed a high pressure, precision roller compactor to further support the development of spray-dried dispersions. -
News Shire granted EU conditional marketing authorisation for Natpar for the treatment of chronic hypoparathyroidism
Natpar is the first and only licensed recombinant human parathyroid hormone therapy for chronic hypoparathyroidism. -
News Fresenius Kabi to strengthen and diversify product portfolio by acquiring Akorn and Merck KGaA’s biosimilars business
Transactions provide access to attractive pharmaceutical growth markets. -
News Regeneron and Sanofi receive FDA approval of a new once-monthly dosing option for Praluent Injection
Monthly dosing schedule now approved in both US and EU. -
News A new lipid technology for antiseptic emollient products
Formulations can be delivered as spray, drop, solution, medical wipe or dermal patch.and offer non-touch applications. -
News Novartis expands global collaboration with Amgen to commercialize first-in-class AMG 334 program in migraine prevention
Novartis and Amgen to co-commercialize AMG 334 (erenumab) in the US; Novartis to gain exclusive rights in Canada, -
News Sartorius group invests $100 million in expansion of its plant in Yauco
Is the largest single investment Sartorius Group has made outside Goettingen in the current year. -
News Allergan granted marketing authorization by FDA for TrueTear
The first intranasal neurostimulating device proven to temporarily increase tear production. -
News Marken Launches 24 Hour Patient Call Center
Marken has launched a 24 hour, 7 day a week call center for patients enrolled in clinical trials. The call center, known as Marken’s Patient Communication Center, is based in Philadelphia (USA) and is dedicated to the logistics needs of patients who pa... -
News Web monitoring for capturing real-world feedback on drugs
First live demo of PLG’s powerful new AI-based social media listening solution, PLG inVolv, designed to help life sciences firms meet their pharmacovigilance obligations. -
News Sanner passes 60 million euro mark
Significant investments in Bensheim headquarters; consistent portfolio additions; cleanroom expansion in China.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance














































.jpg)